*Free registration is required to use the toolkits provided within HIPxChange. This information is required by our funders and is used to determine the impact of the materials posted on the website.
About the training
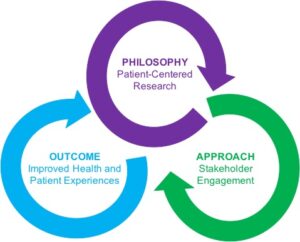
The Patient-Centered Outcomes Research Institute (PCORI) defines “engagement in research” as:
“The meaningful involvement of patients, caregivers, clinicians, and other healthcare stakeholders throughout the research process—from topic selection through design and conduct of research to dissemination of results. PCORI believes that such engagement can influence research to be more patient centered, useful, and trustworthy and ultimately lead to greater use and uptake of research results by the patient and broader healthcare community.” -PCORI, What We Mean by Engagement
This online training is designed to equip researchers to deeply engage patient stakeholders in research. Beyond advisory boards, this training explores multiple ways to engage patient stakeholders, including expanding roles of patients on the research team.
The training is a portal to existing resources and an independent source for high quality, vetted information that has been synthesized from peer-reviewed articles on engagement, stakeholder mapping, marketing, and ethical issues in research.
It offers practical and actionable materials to clear barriers to engagement, including templates to download, videos, resources, and literature references.
It is particularly relevant for researchers focusing on improvements to services that actively involve patients and/or offer shared decision-making opportunities, or those engaging patients in sensitive topics.
“Bringing this [patient] expert insight to individual research projects could make them better: more relevant to patients, more likely to be funded, more likely to recruit participants, and in the end more likely to improve clinical practice and patient experience.” -Researcher on Healthtalk.org
Who should use this training?
This training is designed for researchers and research assistants who want to deeply engage stakeholders across all phases of their research project, beginning at the pre-planning stage through dissemination.
It is designed to support investigators in efforts to expand patient/stakeholder engagement to a higher degree of interactivity and influence.
Learning objectives
 Upon completing this training, the learner will be able to:
Upon completing this training, the learner will be able to:
- Describe a flexible, continuous approach to stakeholder engagement that maps who, when, and how to engage
- Outline principles and best practices for engagement
- Articulate pathways and tools to identify stakeholders, including community partners
- Employ tools and tips to develop an engagement plan, including a timeline and budget
- Describe the value of branding, marketing and social media tools to engagement efforts in a research project
- Identify legal, ethical and IRB considerations relevant to engagement
- Use dissemination as an ongoing engagement strategy
Training content
This training contains 8 stand-alone modules that are designed to be used as you move through the research life cycle. The modules can be used in any order.
- Introduction to the Training
- What, Why and Who of Stakeholder Engagement
- How and When to Engage: Approaches and Timing Considerations
- Preparing Stakeholders and Researchers for Deep Engagement
- Branding, Marketing, and Social Media as Engagement Strategies
- The Role of Research Networks and Communities in Engagement
- Legal, Ethical, and IRB Considerations for Engagement
- Celebration and Compensation for Deeply Engaged Stakeholders
The training also includes Terminology and Frequently Asked Questions sections. Worksheets, guidance documents, articles, and other resources are available for download throughout the training.
Related materials
Other trainings and tools for patient and stakeholder engagement are located on the Browse Toolkits page under the “Patient Engagement” heading.
Registering for the training
After you register for the training, you will need to be manually added to the course. You will be notified as soon as you have access to the course with instructions on how to view it. If you are not a University of Wisconsin student or faculty or staff member, then this process may take a few days.
Development of the training
With funding from a NIH Clinical and Translational Science Award to ICTR and the Wisconsin Partnership Program in the UW School of Medicine and Public Health (SMPH), this course was designed by Sarah Davis, JD, MPA from the Center for Patient Partnerships with significant contributions and editing by Dr. Nancy Pandhi, MD, MPH, PhD It also contains contributions from other SMPH and School of Pharmacy (SOP) researchers, specific content experts, and patients and caregivers who serve on a Patient Advisory Committee.
The course has been reviewed by the Advisory Committee members, research assistants, and engagement experts; and was piloted by several SMPH and SOP researchers and research assistants. More detail available on the Acknowledgements page of the training.
Questions and consultations
Sarah Davis at the Center for Patient Partnerships, a UW ICTR Community-Academic Partnerships (ICTR-CAP) program, is also available to support engagement efforts in your project planning or research. More information about the Center for Patient Partnerships is available here.
If you have questions, comments, or suggestions, or wish to consult on a specific project, please contact Sarah Davis at sarah.davis@wisc.edu